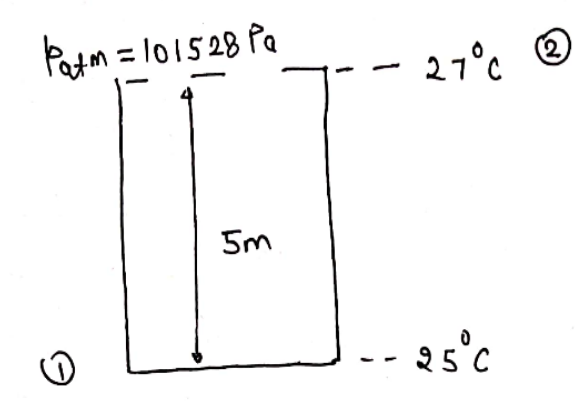
T1=25+273=298KT2=27+273=300Kp2=patm=101528Pa=101528N/m2
Pressure at bottom due to depth of fluid
p=ρghp=103×9.81×5=49050N/m2
Total pressure at bottom
p1=patm+p=101528+49050=150578N/m2T1p1V1=T2p2V2298150578×V1=300101528×V2V2=101528150578×298300×V1V2=1.4929V1=V11.4929V1−V1×100=49.295%

