Answer on Question #72721, Engineering / Mechanical Engineering
A piston cylinder contains gas initially at 3500kPa with a volume of 0.03 cubic meter. The gas is compressed during a process where pV raise to 1.25=C to a pressure of 8500kPa. The heat transfer from the gas is 2.5kJ. Determine the change in internal energy, neglecting changes in kinetic and potential energies. Graph a PV and TS Plane.
Solution:
The polytropic process is one in which the pressure-volume relation is given as
pVn=constant
In our case,
pV1.25=constantp1V11.25=p2V21.25
Thus,
V2=V1(p2p1)1.251=0.03(85003500)1.251=0.01475m3
The work done during the polytropic process is found by substituting the pressure volume relation into the boundary work equation. The result is
W=∫12pdV=1−np2V2−p1V1
So,
W=1−1.258500×103×0.01475−3500×103×0.03=−81500J
Then, according to the first law of thermodynamics,
ΔU=Q−W
where ΔU is the change in the internal energy of the system and W is work done by the system.
ΔU=−2500+81500=79000J=79kJ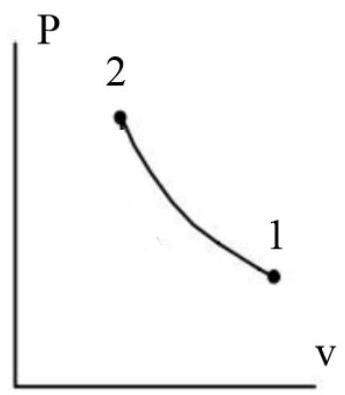
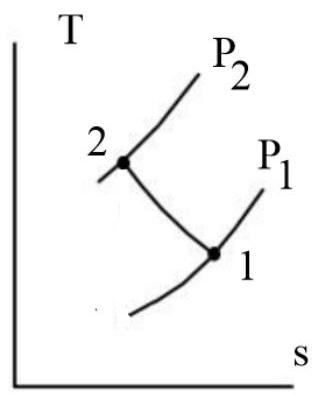
Answer: ΔU=79kJ
Answer provided by AssignmentExpert.com

