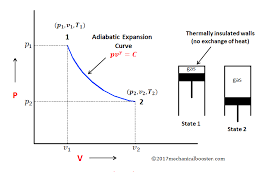
From the above PV2=C where C is constant
1 bar = 105N/m2
from the above p1 = 6.9×105N/m2
Work done (W)=
W=∫v1v2p∂v
W=∫v1v2v2C∂v=C∫v1v2v2∂vC[−2+1v−2+1]from v2→v1
C=[v11−v21]
C=pv2=p1v12=6.9×105N/m2×0.05m2
=34500
Substituting the values of v1,v2 and C in the equation above we get
W=34500[0.051−0.081]N/m
Work done =258750 N/m