Answer on Question #65269 -Physics - Molecular Physics - Thermodynamics
Condition:
**Question 01:**
2.0g of Nitrogen gas is at 270C in a fixed volume. If 20% of its total internal energy is due to rotation what is the average velocity of nitrogen molecules?
What will be the temperature 2.0 g of Helium gas in a fixed volume if the average velocity of its molecules is 20 m/s ?
Plot schematic of Cv v/s T curve based for nitrogen and helium gas based on Kinetic theory.
**Question 02:**
200g of melting ice is introduced to a huge lake at 270C. Find the total entropy change of ice-lake system.
200g of melting ice is introduced to a huge lake at 0.20C. Find the total entropy change of ice-lake system.
Can you draw any conclusion based on result? (Hint: tends to reversible, irreversible etc)
Solution:
**Question 01:**
Nitrogen gas is diatomic gas so N2 has 5 degrees of freedom so total internal energy is U=25kT∗N, where k is Boltzmann constant (k=1.38×10−23J/K), T=270C=543K, N is quantity of molecules.
N=MmNA, where m=2.0g, M is molar mass (Molecular Weight):
M(N2)=28molg, NA is Avogadro constant (NA=6,023∗1023mol−1).
N=282∗6,023∗1023=0,43∗1023=4,3∗1022U=25∗1,38∗10−23∗543∗4,3∗1022=805,54J
If 20% of its total internal energy is due to rotation then 80% is the average energy of motion:
E=0,8∗805,54=644,4J
On the other hand: N∗E0, where N is quantity of molecules and E0 is kinetic energy of one molecule.
E0=NE=4,3∗1022644,4=149,86∗10−22=1,5∗10−20J
On the other hand: E0=m02V2, where V is the average velocity, m0 - molecular mass:
m0=28∗1,66∗10−27=4,65∗10−26kgV=(m02E0)1/2=(4,65∗10−262∗1,5∗10−20)1/2=(0,645∗106)1/2=0,803∗103sm=803sm
Helium gas is monoatomic gas so E0=23kT=m02V2→T=m03kV2
m0=4∗1,66∗10−27=6,64∗10−27kgT=6,64∗10−27∗3∗1,38∗10−23202=641,5∗10−4K=0,064K=−273,086CCv=δTδQ=δTδU→Cv(N2)=δTδ(25kT)=25k;Cv(He)=δTδ(23kT)=23k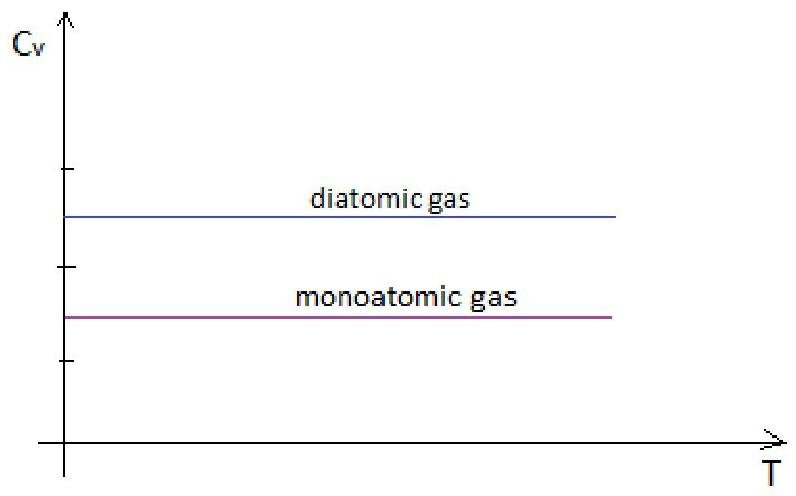
Question 02:
a) Ice->water->steam:
Heat of fusion for ice: q=335kJ/kg . Warmthwhichisnecessaryforicethawing: Q=q∗m=335∗0,2=67kJ .
ΔS1=T0Q=27367=0,245KkJ=245KJ
Heat capacity for water: c=4,187kJ/kg∗ K. Warmthwhichisnecessaryforheatingwaterfrom T0=0C (273K) to T=100C (373 K):
Q=c∗m∗(T−T0)=4,187∗0,2∗100=83,74kJΔS2=Q∗lnT0T=83,74∗ln(273373)=25959KJ
Heat of vaporization for water: L=2260kJ/kg. Warmth which is necessary for water vaporization: Q=Lm=2260∗0,2=452kJ.
ΔS3=TQ=373452=1211,8KJ
Heat capacity for steam: c=2kJ/kg×K. Warmth which is necessary for heating steam from T=100C (373K) to T1=270C (543 K):
Q=c∗m∗(T1−T)=2∗0,2∗170=68kJΔS4=Q∗lnTT1=68∗ln(373543)=29172KJΔS=245+25959+1211,8+29172=56587,8KJ−entropy change for ice.
Theakeloseswarmth: Q=−(67+83,74+452+543)=−1145,74kJ. But temperature of the large lake is a constant so entropy change for lake: ΔS=TQ=543−1145,74=−2110KJ
The total entropy change of ice-lake system: ΔS=56587,8+(−2110)=54477,8KJ
b) Heat of fusion for ice: q=335kJ/kg. Warmth which is necessary for ice thawing: Q=q∗m=335∗0,2=67kJ. ΔS1=T0Q=27367=0,245KkJ=245KJ
Heat capacity for water: c=4,187kJ/kg×K. Warmth which is necessary for heating water from T0=0C (273K) to T=0,2C (273,2 K): Q=c∗m∗(T−T0)=4,187∗0,2∗0,2=0,167kJ
ΔS2=Q∗lnT0T=0,167∗ln(273273,2)=0,12J/KΔS=ΔS1+ΔS2=245,12KJ−entropy change for ice.
Theakeloseswarmth: Q=−(67+0,12)=−67,12kJ. But temperature of the large lake is a constant so entropy change for lake: ΔS=TQ=273,2−67,12=−245,68KJ
The total entropy change of ice-lake system: ΔS=245,12+(−245,68)=−0,56KJ
If we don't take in attention of heating of water, the total entropy change of ice-lake system is 0KJ.
The total entropy of an isolated system always increases over time therefore it is irreversible process.
**Answer:**
Question 01: average velocity of nitrogen molecules is 803sm; temperature of Helium gas is 0,064K=−273,086C
Question 02: The total entropy change of ice-lake system is 54477,8KJ and 0KJ


Comments
Dear hash, what part do you mean?
Part c ?