Properties:
The properties of air at room temperature are
R=0.287kg⋅KkPa⋅m3
cp=1.005kg⋅KKJ
cv=0.718kg⋅KkJ
k=1.4
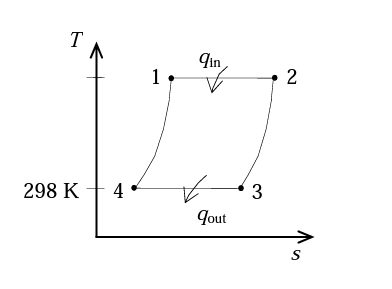
Analysis: Applying the ideal gas equation to the isothermal process -4 gives
P4=P3V4V3=(50kPa)(12)=600kPa
Since process 4-1 is one of constant volume
T1=T4(P4P1)=(298K)(600kPa3600kPa)=1788K
Adapting the first and work integral to the heat addition process gives
qin=W1−2=RT1lnV1V2=(0.287kg∗KKJ)(1788K)ln(12)=1275kgKJ
Similarly
qout=W3−4=RT3lnV3V4=(0.287kg∗KKJ)(298K)ln(121)=212.5kgKJ
The net work is then
Wnet=qin−qout=1275−212.5=1063kgKJ

