Answer on Question #58182 – Chemistry – Organic Chemistry
Task:
Assume that 3.0g of aniline and 4.5mL of acetic anhydride (density ρ=1.08gmL−1) are used in the preparation of acetanilide.
1) What is the limiting reagent? Justify your answer.
2) What is the theoretical yield in grams of acetanilide?
3) What is the % yield if 3.30g of acetanilide are obtained?
Solution:
We write the reaction of interaction the aniline with acetic anhydride to form acetanilide:
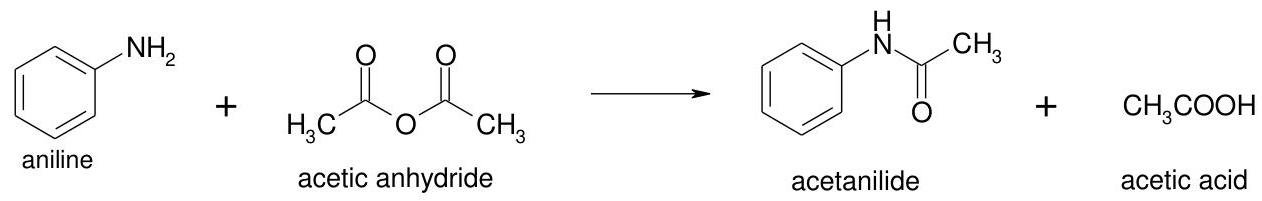
We find the molar mass of aniline and acetic anhydride:
M(aniline)=M(C6H5NH2)=12×6+1×7+14×1=93(molg);M(acetic anhydride)=M(C4H6O3)=12×4+1×6+16×3=102(molg).
Convert each substance to moles:
n(aniline)=M(C6H5NH2)m(C6H5NH2)=93g/mol3g=0.0322mol;n(C4H6O3)=M(C4H6O3)m(C4H6O3)=M(C4H6O3)ρ(C4H6O3)×V(C4H6O3)=102g/mol1.08g/ml×4.5ml=0.0476mol
1) To find the limiting reagent, take the moles of each substance and divide it by the coefficient of the balanced equation. The substance that has the smallest answer is the limiting reagent.
Determine the limiting reagent:
For aniline: 0.0322/1=0.0322
For acetic anhydride: 0.0467/1=0.0476
There is enough acetic anhydride to produce 0.0467 mol acetanilide, but only enough aniline to produce 0.0322 mol acetanilide. This means that the amount of acetanilide actually produced is limited by the aniline, which is therefore the limiting reagent.
Answer:
Limiting reagent is aniline.
2) Aniline and acetanilide stand in a one-to-one molar relationship, so 0.0322 mol of aniline produces 0.0322 mol of acetanilide. The amount of acetic anhydride does not play a role, since it is in excess.
Then,
n(aniline)=n(acetanilide)=0.0322mol.
We find the molar mass of acetanilide:
M(acetanilide)=M(C8H9NO)=12×8+1×9+14×1+16×1=135(molg).
We find the theoretical yield in grams of acetanilide:
m(acetanilide)=n(C8H9NO)×M(C8H9NO)=0.0322mol×135molg=4.347g≈4.35g.
Answer:
The theoretical yield of acetanilide is 4.35g.
3) What is the % yield if 3.30 g of acetanilide are obtained?
Theoretical yield: 4.35g.
Actual yield: 3.30g.
Percent yield (%Yield):
%Yield=Theoretical Yield (mass or moles)Actual Yield (mass or moles)×100%.
Then,
Percent Yield=Theoretical YieldActual Yield×100%=4.35g3.30g×100%=75.86%.
Answer:
Percent yield of acetanilide is 75.86%.
http://www.AssignmentExpert.com/

