Answer on Question #85704 – Chemistry – Inorganic Chemistry
[Co(H2O)6]2+ ion is more stable than [Co(H2O)6]3+ ion justify it?
Solution:
[Co(H2O)6]2+ is more stable than [Co(H2O)6]3+ because Co2+ is more stabilised by weak field ligand, H2O than is Co3+ .

With weak-field ligands, the half-filled shell with all spins parallel has extra stability. It's the same reason that Cr has electron configuration [Ar] 4s13d5 rather than [Ar] 4s23d4 .
Co2+(3d7)=(t2g)5(eg)2 has higher value of crystal field stabilization energy than Co3+(3d6)=(t2g)4(eg)2 in the weak octahedral field leading to greater stability of Co2+(aq) than Co3+(aq) .
On the other hand, [Co(H2O)6]3+ is more stable than [Co(H2O)6]2+ because Co3+ is more stabilised by strong field ligand NH3 than is Co2+ . This is due to higher value of crystal field stabilization energy for Co3+(3 d6)=(t2 g)6(eg)0 than Co2+(3 d7)=(t2 g)6(eg)1 in the strong octahedral field ligand NH3 leading to greater stability in former. The crystal field diagram is then:
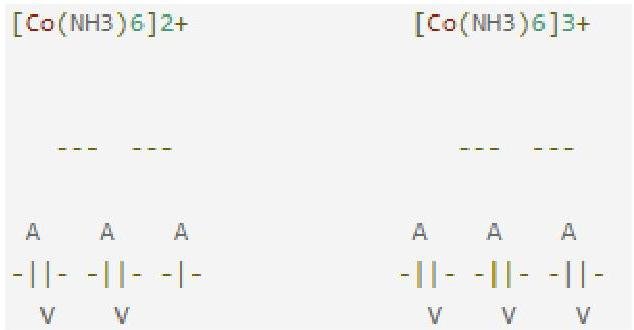
Note that the full orbital level also has extra stability.
Answer provided by www.AssignmentExpert.com

