step 1: calculate the molar mass of solute.
molar mass of solute = (12.01 g/mol × 12) + (1.008 g/mol × 22) + (16.00 g/mol × 1
molar mass of solute = 342.296 g/m
step 2: calculate the number of moles of solute.ol1)
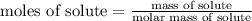
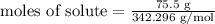
moles of solute = 0.22056933 mol
Step 3: Calculate the molality of solution.
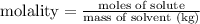
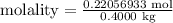
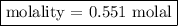
Comments