It is given that burning one mole of caffeine gives heat.
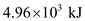
The given mass of caffeine is 5g.
Molar mass of caffeine can be calculated as shown below.
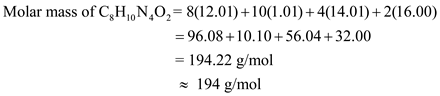
Calculate the number of moles of caffeine burned as follows:

Q = mc ΔT
0.0258 ×4.96 × 103 × 284.37
= 3.64 × 104J/°C
Comments