Ans:-
Antoine equation can be used to determine the vapor pressure of the substance,
p=10(A−T+CB)
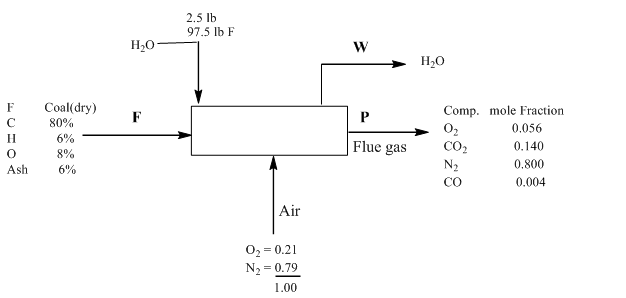
Since the coal contained the 2.5 %moisture and the basis is 100lbmol , the water or moisture entering the system can be calculated as follows:
H2Oin=2.5
=1002.5×100
=2.5mol in F
Therefore, the coal entering the system is
Coalin=100−2.5
=97.5mol in F
Further, F and A can be determined using material balances as follow:
Cin=80%=0.80mol
Cout=CO2(out)+COout
=14%+0.4%=0.140+0.004=0.144mol
Thus, to determine F
0.80F×121=(0.144)×(100)0.080F=14.4F=18lbmol
The molar mass of the carbon is12lb/mol . So,
Mass of F= Number of moles× Molar mass
=18lbmol×12lb/mol=216lb
Nitrogen
N2(in)=79%=0.79molN2(out)=80%=0.80mol
Thus, to determine A
80=(A)×(0.79)A=101.26lbmol
Now, the moles of hydrogen in dry cola and water is determined as follow:
Moles of H in dry coal=(MolarmassofHMassofHinF)
=12.86moles
From the ideal gas equation we find the temperature of Coal gas
T=n×RP×V=12.86×62.36329.9×100=3.728K
Dew point will be 12


Comments